Aggregation-Induced Enhancement of Deep-UV Response in Indoloindole -Based Organic Photodetectors (Adv. Optical Mater. 2025, e02481 ; DOI:10.1002/adom.20250248)
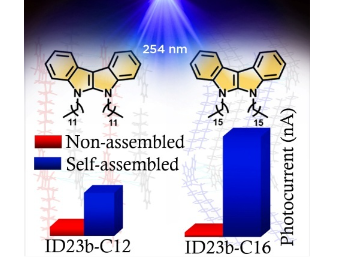
Organic photodetectors operating in the deep-ultraviolet region are crucial for environmental monitoring, biomedical imaging, and sterilization systems. We report two novel indolo[2,3-b]indole derivatives with systematic alkyl chain variation: 5,6- didodecyl-5,6-dihydroindolo[2,3-b]indole (ID23b-C12) and 5,6-dihexadecyl-5,6- dihydroindolo[2,3-b]indole (ID23b-C16). Both compounds exhibit wide bandgaps (>3.5 eV), high thermal stability (decomposition temperature >410 °C), and form aggregates in water:THF mixtures with distinct morphological characteristics. Using simple planar FTO/molecule/FTO devices, films prepared from pre-aggregated solutions achieve photoresponsivities of 32.5 mA/W (ID23b-C12) and 74.8 mA/W (ID23b-C16) at 254 nm under 2 V bias, representing 6.1× and 20.8× enhancements compared to molecularly dispersed films. Detectivity values reach 2.8×10¹⁰ Jones and 1.3×10¹¹ Jones, respectively. This single-component approach achieves performance competitive with complex donor-acceptor heterojunctions, demonstrating solution-state molecular organization as an effective strategy for efficient deep-UV photodetection without elaborate device engineering.
- Research Area :Chemical Sciences & Technology Division (CSTD)

