Accessing Isomers of Benzothienonaphthofurans via Regio Controlled Structural Fusion of Electrophilic Benzothiophenes with Naphthols (J. Org. Chem. 2025, 90, 16697–16710; DOI: 10.1021/acs.joc.5c01703)
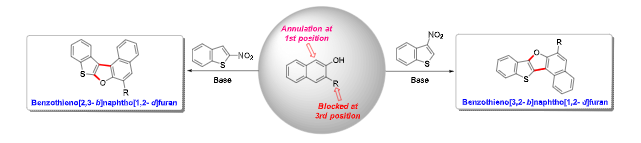
Polycyclic aromatic compounds have found immense applications as speciality chemicals and in materials and devices. In continuation of our pursuit of accessing heteroacenes, we designed reactions for accessing hitherto unknown or seldom explored isomers of benzothienonaphthofurans. We have found that a base-mediated regio-controlled annulation of electrophilic benzothiophenes with 3- substituted-2-naphthols would furnish heteroacenes, namely benzothieno[3,2- b]naphtho[1,2-d]furan and benzothieno[2,3-b]naphtho[1,2-d]furan. This was made possible by blocking the 3 rd position of 2-naphthol (most favourable position for annulation), which in turn forces the annulation to happen from the 1 st position. This synthetic methodology for accessing isomers of benzothienonaphthofurans proved to be broadly applicable to various 3-substituted-2-naphthols and electrophilic benzothiophenes. Its scalability was demonstrated through gram-scale synthesis, and the fundamental photophysical properties of the products were also investigated.
- Research Area :Chemical Sciences & Technology Division (CSTD)

